Virtual Consultations Available
Our team wants anyone who's interested in hair transplantation to be able to attend a consultation. We offer free virtual consultations over FaceTime, WhatsApp, and Zoom so patients outside Houston—or those who have busy schedules—can conveniently explore their options with Dr. Arocha himself.
Welcome to Arocha Hair Restoration Leading Hair Transplant Specialist in Houston
Since 2002, Bernardino A. Arocha, M.D. has been providing patients throughout Houston and beyond with customized hair restoration procedures and truly transformative results. His interest in hair restoration began in his 20s when he started to experience hair loss himself. He understands the psychological and emotional toll hair loss can have on an individual, which is why he has dedicated his career to helping patients experience the same elevated quality of life that hair transplantation has provided him.
If you've noticed thinning or balding and want to address it with the help of a board-certified surgeon, we can help you explore your options. Using his artistic eye and detail-oriented approach, Dr. Arocha can bring back your confidence and elevate your quality of life with a fuller head of hair.
Words From Our Doctor Himself
Understanding His Philosophy
"Throughout my professional career I have maintained an unwavering commitment to hair restoration. Watching my patients’ self-esteem soar and listening to my patients express their gratitude for changing their lives has had a profound impact on my practice. I treat every patient with dignity and respect, and work tirelessly to assure my patients that they are receiving my best surgical effort. If you are willing to place your trust in me, I promise that I will do my best to give you the best customized hair replacement results possible. I would consider it an honor to help you reach your hair restoration goals."
- Bernardino A. Arocha, M.D.
"I cannot wait to see the results from my recent procedure and continue working with Dr. Arocha in the future!"
First, I must say a very big thank you to Dr. Arocha for his humility and dedication to work, he listened to me and helped me on this journey… my experience was very exceptional and i am very happy for the results I am already seeing, it’s great. Thank you
View On GoogleI walked into Arocha Hair Restoration demoralized by my progressive hair loss. I met with Dr. Arocha and his team and was filled with hope afterwards. From his system, I went from a non-candidate to a candidate for an FUE procedure! The staff is extremely helpful and friendly-- they even bought me lunch during our break. I cannot wait to see the results from my recent procedure and continue working with Dr. Arocha in the future!
View On GoogleTransform Your Hairline and Quality of Life Contact Our Houston Office Today
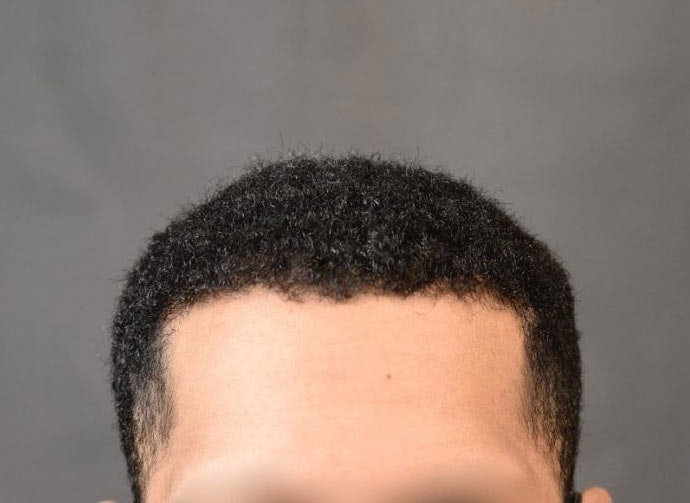
Balding and thinning can cause feelings of self-consciousness for men and women alike. Fortunately, there are plenty of ways to help you achieve the full head of hair you desire. Combining the latest hair transplantation techniques with an artistic eye, Dr. Arocha has transformed the lives of many patients.
Whatever the cause or extent of your hair loss, we're confident we can help. Request your consultation today by calling our office in Houston:
(713) 526-4247
Our Patient Explains Why He Loves His Results
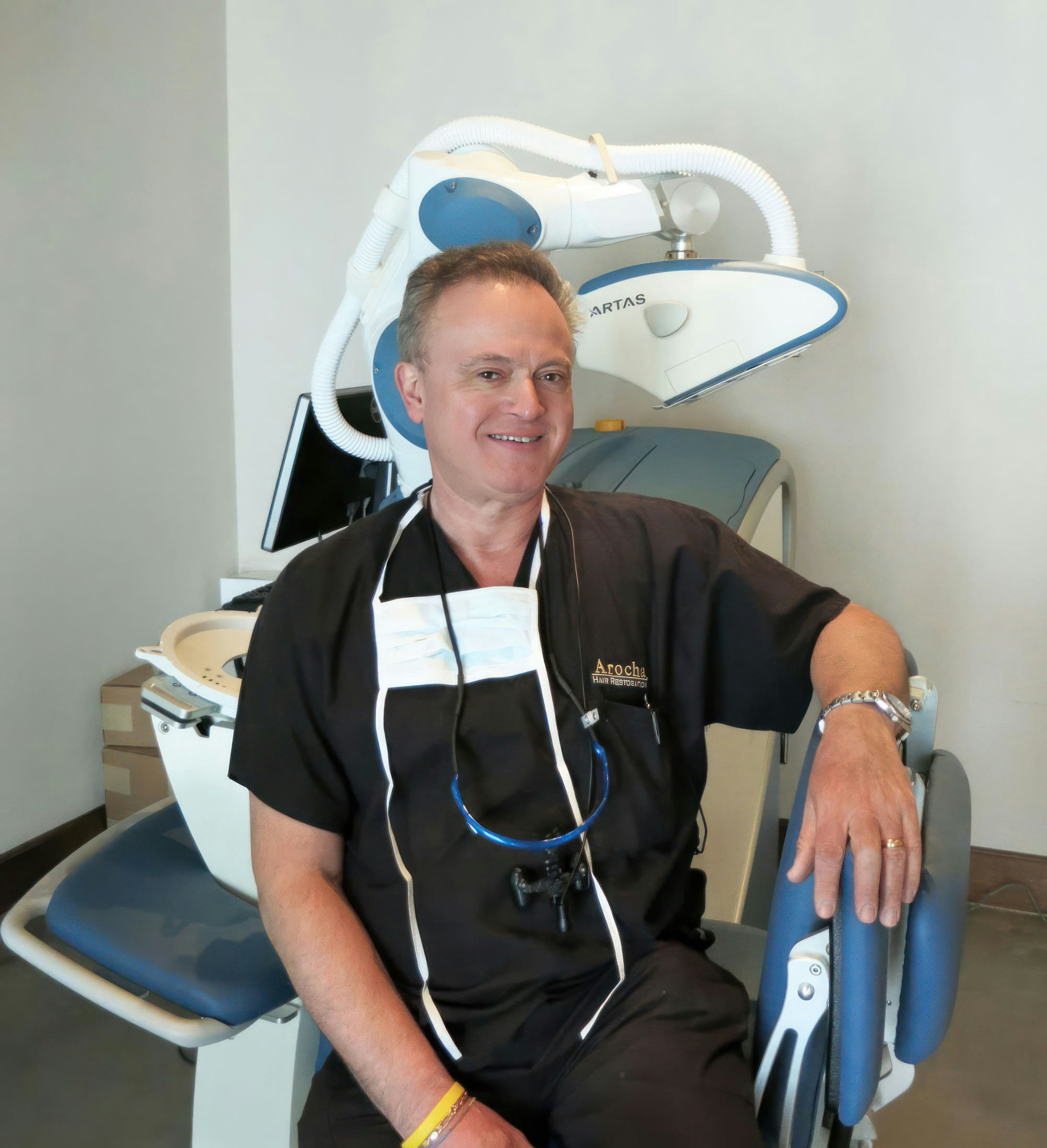
Meet Bernardino A. Arocha, M.D.
A fellow of the International Society of Hair Restoration Surgeons and a diplomate of the American Board of Hair Restoration Surgery, Dr. Arocha wants to partner with you to improve your hairline and thicken your hair. He has experienced firsthand how hair restoration can transform a person's life, mental health, and emotional well-being, and he wants to give you the same kind of results he received. He believes that together, you can achieve natural-looking hair restoration results that last.

Featured in the Media
Dr. Arocha is a leader in his field and is known for developing his own hair transplantation techniques for the most natural-looking results. He has been featured in the media on many occasions, and you may have encountered his work in one of several popular magazines or TV shows.
Hair Transplant Results of Our Featured Services on Our Real Patients
Our Complete List of Services
- ARTAS® Robotic Hair Transplant
- Beard Transplant
- Corrective (Revision) Hair Transplant Surgery
- Ethnic Hair Restoration
- Eyebrow Transplant
- Eyelash Restoration
- Facial Hair Transplants
- Follicular Unit Extraction (FUE)
- Follicular Unit Transplantation (FUT)
- Hair Loss Treatment
- LaserCap® LCPRO™ Hair Loss Treatment
- Nonsurgical Hair Restoration
- Scalp Micropigmentation
- Transgender Hair Transplants
Paying for Your Hair Restoration
While insurance won't contribute to the cost of your hair restoration, we are proud to work with CareCredit®, a third-party lender that allows qualifying patients to pay for their treatment in affordable monthly installments.
Patients Near and Far Travel to Our Houston Office
First, I must say a very big thank you to Dr. Arocha for his humility and dedication to work, he listened to me and helped me on this journey… my experience was very exceptional and i am very happy for the results I am already seeing, it’s great. Thank you
View On GoogleI walked into Arocha Hair Restoration demoralized by my progressive hair loss. I met with Dr. Arocha and his team and was filled with hope afterwards. From his system, I went from a non-candidate to a candidate for an FUE procedure! The staff is extremely helpful and friendly-- they even bought me lunch during our break. I cannot wait to see the results from my recent procedure and continue working with Dr. Arocha in the future!
View On Google